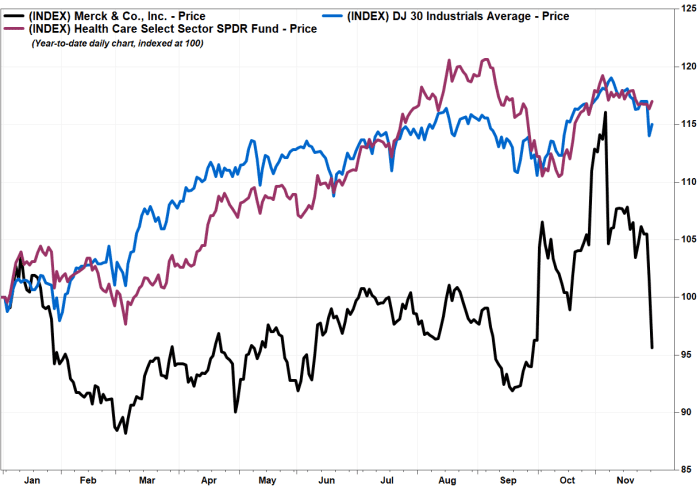
Shares of Merck & Co. Inc. sank Monday, to buck the gains in the wellness care sector and the broader stock market, immediately after Citi Investigation backed away from its bullish stance on the drug maker, citing problems around its HIV and COVID-19 therapies.
The stock
MRK,
fell 5.7% in afternoon buying and selling towards a two-thirty day period reduced. The inventory is the most significant decliner amongst the components of the Dow Jones Industrial Typical
DJIA,
which ran up 305 details, or .9%, and of the SPDR S&P Well being Treatment Pick Sector exchange-traded fund
XLV,
which rallied .6%.
The inventory has now tumbled 17.6% given that closing at a record $90.54 on Nov. 4.
Citi’s Andrew Baum slice his rating to neutral, following staying at purchase for at the very least the past 2 1/2 decades. He slash this inventory cost concentrate on to $85 from $105.
Baum mentioned his “long-standing” bullish thesis on Merck was based mostly on the “under-appreciation” of the company’s drug pipeline, particularly its HIV treatment method islatravir, which he expected would offset the coming decline of exclusivity of its blockbuster most cancers treatment Ketruda. Nevertheless, he no extended expects any revenue from islatravir.
“We location a higher chance that [Merck] will abandon islatravir development in the following three months specified probable superior regulatory issues,” Baum wrote in a observe to consumers.
Baum explained he removed all estimates for islatravir from his fiscal models, next the company’s announcement previously this month that a dose-dependent lessen in lymphocyte counts was noticed in a Period 2 trial. He reported that indicates its extremely most likely that materially higher doses demanded would lead to “diverse and unacceptable” adverse activities.
On Nov. 18, Merck explained it stopped dosing in the demo, and on Nov. 23, the corporation introduced a “temporary pause” in enrollment in the Period 2 review.
Merck’s stock has get rid of 9.8% because Nov. 18.
“We anticipate the diminishing outlook for islatravir to additional expedite Merck’s business enhancement initiatives,” Baum wrote. “We open a detrimental catalyst check out on the inventory along with today’s report.”
FactSet, MarketWatch
But disappointment around islatravir is not Merck’s only dilemma.
Baum reported the clinical profile of Merck’s antiviral to take care of COVID-19, Lagevrio, carries on to deteriorate, placing estimates for Lagevrio for following year and past at “material risk.” He explained it was “obvious” to him from inception that Lagevrio would have a hazard analysis and mitigation method (REMS) drug security application expected by the Meals and Drug Administration for the reason that of danger of start problems.
Really do not miss MarketWatch’s day-to-day “Coronavirus Update” column.
“Since that time, we have learnt that Lagevrio’s efficacy is materially decrease than that claimed by possibly monoclonals these types of as Regeneron’s Ronapreve as effectively as Pfizer’s Paxlovid (on an interim assessment),” Baum wrote.
He also believes it is very likely that resistance will emerge about time to monotherapy Lagevrio usage.
“We hope the Fda to propose monoclonal antibodies to be most well-liked remedy in immunocompromised individuals in purchase to cut down the threat of resistance,” Baum wrote.
As a final result, he stated the Food and drug administration could conclude up restricting Lagevrio acceptance to only non-vaccinated or immunocompromised clients.”
Merck’s stock has dropped 4.3% year to day, while shares of Regeneron Pharmaceuticals Inc.
REGN,
have climbed 36.1% and of Pfizer Inc.
PFE,
have hiked up 44.2%. In the meantime, the SPDR health and fitness treatment ETF has superior 17.% this yr and the Dow has acquired 15.%.
